 Human fetal neural retinal cells from second-trimester fetuses can be expanded into a large number of undifferentiated cells in vitro and mature retinal cells. Candidate cells for this treatment exclusively express the cone-rod homeobox-containing gene (CRX), but no expression of Opsin (the main chromophore of the mature photoreceptor). This means that the transplanted cells should be photoreceptor precursors before being delivered to patients. 
Subsequent reports suggested that only transplanted cells isolated from fetuses at a specific gestational stage and already committed to a photoreceptor cell fate could facilitate visual recovery. The most relevant previous clinical studies to treat retinal degeneration used fetal retinal sheets and immature neural retinal cells, and there were no obvious signs of immune rejection however, parallel animal studies suggested there was only limited progenitor cell integration into the neural retina. Secondly, transplanting ESCs or iPSCs carries the potential risk of tumor formation and gene mutation. RPCs derived from ESCs or iPSCs require a longer time and more steps to induce in vitro. Firstly, RPCs derived from fetal neural retinas have low immunogenicity and can stably maintain their characteristics over many passages. With regard to the source of RPCs, fetal-derived RPCs may have an advantage for cell therapy. Currently, retinal progenitor cells (RPCs) are usually derived from fetal retinas, embryonic stem cells (ESCs), and induced pluripotent stem cells (iPSCs). These findings suggested that it is possible to transplant human RPCs to treat retinitis pigmentosa patients. .jpg)
Studies using retinitis pigmentosa animal models indicated that the transplanted cells were able to differentiate into photoreceptors, integrate into the host retinas, and rescue vision. Their studies provided the first evidence that hESC-derived cells can be used as a new form of therapy to treat retinal degeneration. reported on mid-term and long-term outcomes when using human embryonic stem cell (hESC)-derived retinal pigment epithelial (RPE) cells to treat dry atrophic age-related macular degeneration and Stargardt's macular dystrophy. Compared to gene therapies, cell transplantation might have good potential to rescue or replace dysfunctional photoreceptors. Therefore, the identification of causative genes must be a prerequisite if gene therapies are to be applied to treat retinitis pigmentosa patients. Retinitis pigmentosa is related to different genetic etiologies, all of which induce the death of photoreceptors. Retinitis pigmentosa is an inherited retinal dystrophy that is characterized by the onset of night blindness, the early loss of peripheral vision, and later the loss of central vision. Retrospectively registered on 5 December 2008. WHO Trial Registration, ChiCTR-TNRC-08000193. Our study for the first time confirmed the long-term safety and feasibility of vision repair by stem cell therapy in patients blinded by retinitis pigmentosa. We observed a significant improvement in visual acuity ( P < 0.05) in five patients and an increase in retinal sensitivity of pupillary responses in three of the eight patients between 2 and 6 months after the transplant, but this improvement did not appear by 12 months.  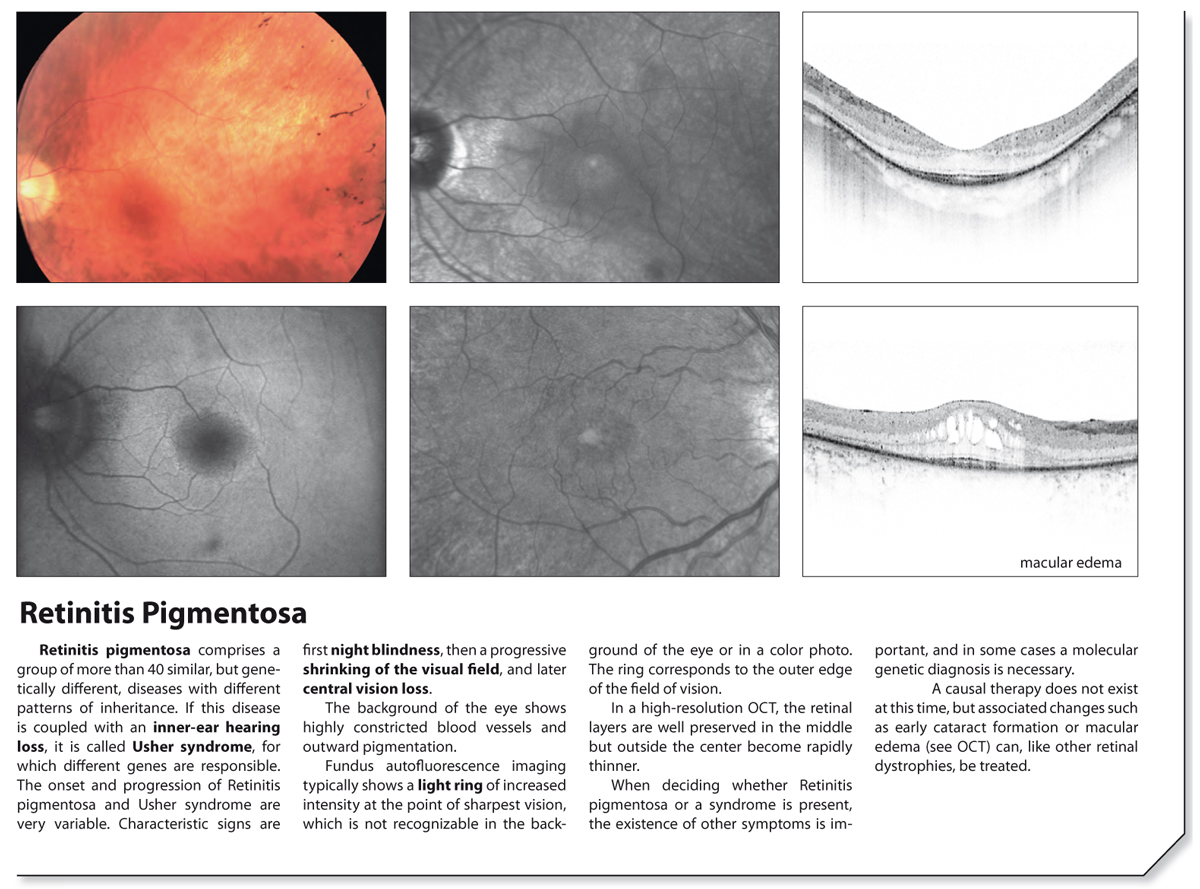
In the retinis pigmentosa patients given RPC injections, we also did not observe immunological rejection or tumorigenesis when immunosuppressive agents were not administered. Most importantly, we did not find tumor formation or immune rejection. ResultsĪfter RPC transplantation in RCS rats, we observed moderate recovery of vision and maintenance of the outer nuclear layer thickness. Based on these results, we conducted a phase I clinical trial to establish the safety and tolerability of transplantation of RPCs in eight patients with advanced retinitis pigmentosa. In our pre-clinical study, purified human fetal-derived retinal progenitor cells (RPCs) were transplanted into the diseased retina of Royal College of Surgeons (RCS) rats, a model of retinal degeneration. However, long-term safety studies and visual assessment have not been thoroughly tested in retinitis pigmentosa patients. Vision preservation was observed in retinitis pigmentosa animal models after retinal stem cell transplantation. Retinitis pigmentosa is a common genetic disease that causes retinal degeneration and blindness for which there is currently no curable treatment available. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
